| MATERIALS AND METHODS |
Hematoporphyrin (Hp) was purchased from Serva, meso-tetraphenylporphine tetrasulphonate (TPPS4) was provided by Porphyrin Products (Logan, UT). Porphyrins were used without further purification. Phosphate buffered saline (PBS) (pH 7.2) solutions were prepared by dissolving the crude sensitizers in 0.02 ml of 0.1 M NaOH followed by dilution with PBS to concentration of 10^(-3) M (stock solution). The stock solutions were kept in the dark at low temperatures. Further dilution with PBS was performed immediately before the measurements.
Human serum albumin (HSA) and bovine serum albumin (BSA) were purchased from Fluka (USA) and Reanal (Hungary). The stock solutions were prepared by dissolving crude HSA or BSA directly in PBS. To prepare samples for illumination the solutions of sensitizer and albumin were mixed at molar ratio 1:1 to produce 2 ml of solution.
Illumination of solutions was performed by exposing 1 cm^2 of the photosensitizer solution to laser light in a quartz cuvette (volume, 2 ml; light path length, 1 cm). An argon ion laser (wavelength, 514 nm, fluence rate not higher than 100 mW/cm^2) and He-Ne laser (wavelength, 632 nm, fluence rate 25 mW/cm^2) were used as a light source.
The absorption spectra of sensitizers solutions were measured with a PC1000 Plug-in Spectrometer (Ocean Optics, Inc., USA).
The efficiency of the photobleaching of a sensitizer is strictly related to the amount of the absorbed light quanta, that is proportional to the fluence of light source, I, and illumination time, t, i.e. to the incident illumination dose, D=I t. The initial absorption bleaching rate constants of the investigated sensitizers in aqueous solutions were evaluated by the following formula [5]:
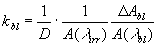
Experiments with ALA-induced protoporphyrin IX were
performed with female hairless BALB/c mice. Animals were anesthesized with
subcutaneous injection of 0.05-0.1 ml of Hypnorum Dormicum 15-30 min before
application of the 5-aminolevulinic acid (ALA) cream. ALA hydrochloride
was kindly supplied by Photocure AS (Oslo, Norway). A cream was prepared
using 20% ALA in an ointment (Unguentum Merck, Germany). Approximately
0.1 mg of the cream was applied topically on a spot of approximately 1
cm^2 and covered with adhesive dressing (Smith and Nephew, England). Animals
woke up within 1 h and appeared normally active. A Perkin-Elmer LS50B luminescence
spectrometer equipped with fiber-optic probe was used for the fluorescence
detection in vivo.
![]()
|
|
|
|